Engineering and Consulting
Services for the Medical
Device Industry and Beyond
HTS accelerates your path to market with specialized engineering and regulatory compliance services for companies developing software and hardware products.

Worldwide
Expertise

In-Person
Collaboration

Proven
Experience

Medical Device
Experts
How We Work




Custom Product Development
Have a product you need built? Whether managing the entire project or supporting a specific sub component, HTS can take your concept from requirements to delivery, handling electronics and hardware architecture, firmware, software, testing, and regulatory compliance.
Fractional CTO Services
For teams that need senior technical leadership, HTS provides fractional CTO support, helping establish system architecture, guiding key technical decisions, mentoring your team, and ensuring your product is built on a solid, scalable foundation.
Engineering Support
If you already have a team but need extra capability, our engineers work directly alongside your staff to accelerate development, solve complex issues, and provide specialized firmware, software, and regulatory compliance expertise.
Technical & Regulatory Document Drafting
Under NDA, our engineers review your product code base and collaborate with your technical team to gain a deep understanding of your product. Combining this knowledge with our regulatory affairs expertise, HTS prepares drafts of technical and regulatory documentation for your team to review – jump starting your path to regulatory approval in key markets.
Our Multidisciplinary Team
The HTS team is a multidisciplinary group of experienced professionals
- Embedded Firmware Engineers
- Application & Cloud Software Engineers
- Medical Device Cybersecurity Engineers
- Software Engineers with a Documentation Focus (510(k) Support)
- Medical Device Regulatory Affairs Specialists
- Medical Device QMS Experts






Our Technology Stack Expertise
Precision engineering for high reliability medical devices and software systems. Trusted by leading healthcare innovators worldwide.

Electronics Hardware
Our team has deep experience with circuit design, circuit simulation, and PCB layout, particularly as applied to medical devices. Whether you're looking for a complete design, an engineering peer review, or help resolving a particular technical challenge - we’re here to help.
- Medical sensing applications (EMG, EKG, pulse oximetry, etc.)
- IOT communications hardware (wifi, bluetooth, cellular)
- Power electronics applications (AC/DC, DC/DC, battery management, load switching (AC & DC), motor controllers)
- Design to ensure compliance with IEC 60601 (basic safety, essential performance, EMC, etc.)

Embedded Firmware
HTS has deep expertise with embedded firmware development. We often assist clients with ‘next level’ challenges: diagnosing the source of mysterious ‘bricking’ incidents, developing interface libraries for un-documented hardware, resolving underlying issues on embedded linux platforms, etc.
C
C++
More
Microprocessors
(Bare Metal & RTOS)
Wireless
Modules
Embedded
Linux
- Firmware design and testing for medical device level reliability, safety, and traceability
- Data acquisition and digital signal processing
- Outbound data transmission via Bluetooth, Wifi, and Cellular networks.
- Sleep state implementation for ultra low power mobile devices
- Extensive experience with embedded linux.

Applications Software
HTS has extensive experience with applications software that interacts with custom hardware. Whether it's medical imaging device control software running on Linux, a Windows desktop app for reviewing device data, or an iOS mobile app that interacts with a medical device - HTS is here to help
C
Qt
C#
Java
More
Linux
Windows
iOS
Android
- Graphical user interfaces and medical imaging device control software
- Desktop programs for reviewing data generated by hardware products
- Configuration management and diagnostic tools for service technicians
- Cross platform app development for mobile apps interacting with physical devices
- Underlying technology and hardware interfaces: device drivers, etc.

Cloud Software
Cloud software is an integral component of many hardware products, particularly when those products are deployed at a substantial scale. HTS has experience developing high reliability cloud software that interacts with large fleets of deployed devices.
Python
JS
React
More
Azure
AWS
GCP
- Safety critical medical alarm reporting and response management software
- Software for receiving, analyzing, and displaying data from large fleets of IOT devices
- Fleet monitoring, configuration, and log retrieval
Medical Device Regulatory Compliance
HTS provides a comprehensive suite of technical and regulatory compliance consulting services for the medical device industry.

Technical Documentation
Whether you need help documenting design records, developing an entire Technical File / DHF, or assistance throughout the 510(k) or notified body auditing process, HTS can assist in developing documentation compliant to each market’s regulations.
Markets
- US - FDA
- EU - MDR
- AUS - TGA
Key Documentation
- Design History File and DMR
- Technical File
- Quality Documentation

510(k) Submissions
We assist clients throughout the entire 510(k) submission process, supporting the preparation of complete and well-structured applications. Our team has particular strength in the software and cybersecurity sections of 510(k) submissions.
Software Documentation
- Software Description
- Software Risk Assessment
- Software Requirements Specification (SRS)
- System and Software Architecture Design Chart
- Software Testing: Verification, Validation, and Traceability
Cybersecurity Documentation
- Cybersecurity Risk Management Report
- Cybersecurity Risk Assessment
- Software Requirements Specification (SRS)
- Cybersecurity Labeling

Cybersecurity Penetration Testing
Our independent cybersecurity penetration testing uncovers vulnerabilities in your product and includes a comprehensive report. We also provide practical recommendations to resolve any issues identified.
Cybersecurity testing methods include:
- Network Security Testing
- Access Control Testing
- Logging and Audit Trail Inspection
- Cryptographic Key Management Review
- Physical Access Exploit Testing
- Fuzz Testing (Firmware and Bootloaders)

Quality Management System Consulting
With extensive experience in ISO 13485 and ISO 9001, HTS supports the development and refinement of quality management systems that are practical, scalable, and compliant with industry requirements. We can assist with your software development life cycle process to ensure compliance with IEC 62304.
Our services include:
- QMS Development and Documentation
- System Review and Independent Auditing
- Targeted Quality Consulting and Advice
- Assistance preparing for Notified Body audits
Notable Engagements
Medical Imaging Scanner
Medical Imaging Scanner
- Engineering architecture
- User interface design
- Cybersecurity testing
- QMS Consulting
- 510K documentation

EMG Sensor and Cross Platform App
EMG Sensor and Cross Platform App
- Fractional CTO services
- Electronics hardware design consulting
- Embedded firmware
- IEC 60601 compliance consulting
- Cross platform mobile app development
Embedded Apps & Cloud Software
Embedded Apps & Cloud Software
- Software system architecture: cellular IOT devices at scale
- Software development: embedded linux apps and cloud software for fleet management, data analysis and display: C++, Python, etc.
- Graphical Display Design
Top Global Talent. In Person Support
With team members across NZ, AUS, Europe, UK, and the USA, we deliver practical expertise and in-person collaboration when projects demand it.
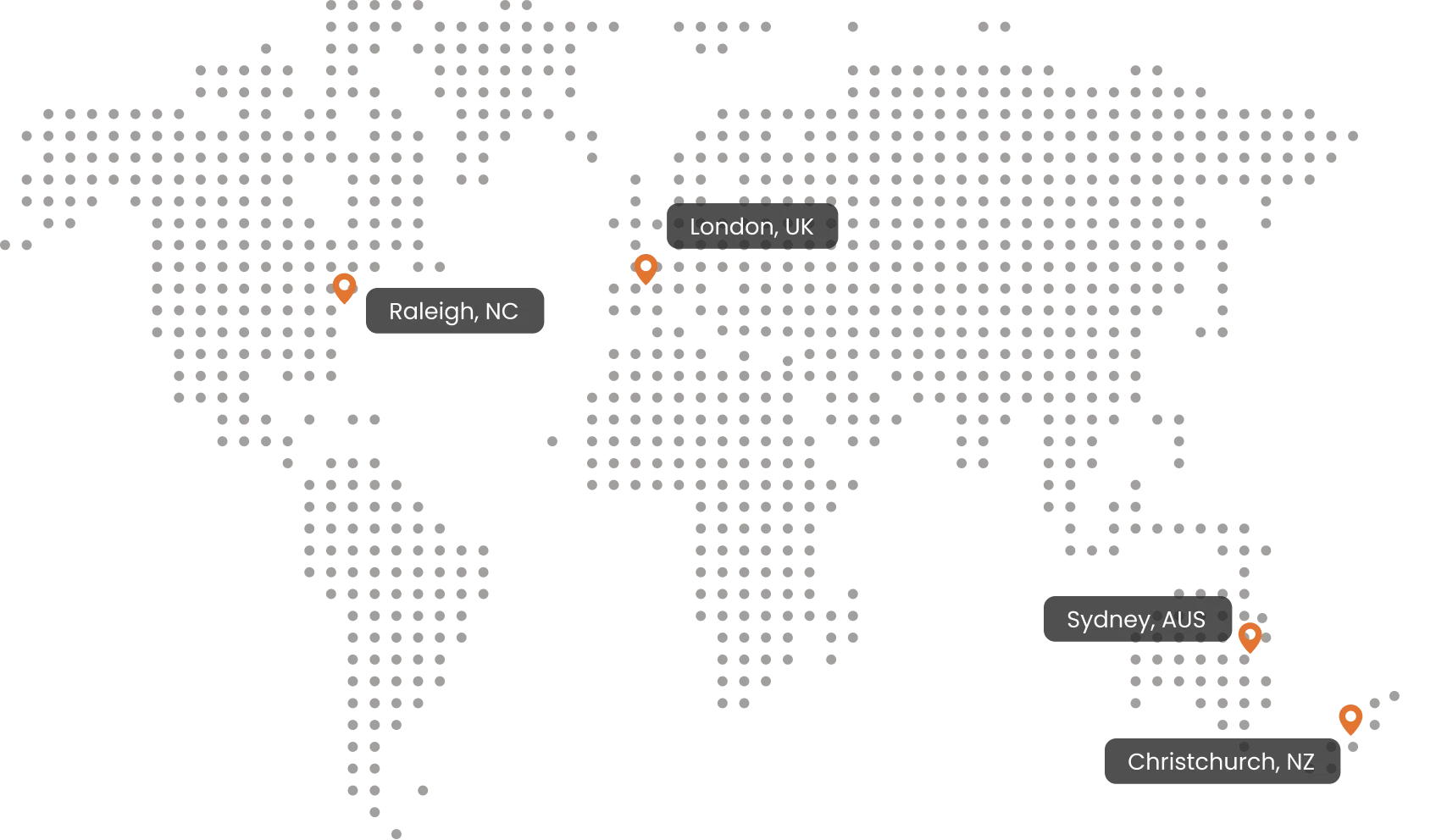

Raleigh, NC
London, UK
Sydney, AUS
Christchurch, NZ
